Myctophidae
Lanternfishes
Melanie L.J. Stiassny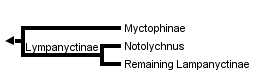

This tree diagram shows the relationships between several groups of organisms.
The root of the current tree connects the organisms featured in this tree to their containing group and the rest of the Tree of Life. The basal branching point in the tree represents the ancestor of the other groups in the tree. This ancestor diversified over time into several descendent subgroups, which are represented as internal nodes and terminal taxa to the right.

You can click on the root to travel down the Tree of Life all the way to the root of all Life, and you can click on the names of descendent subgroups to travel up the Tree of Life all the way to individual species.
For more information on ToL tree formatting, please see Interpreting the Tree or Classification. To learn more about phylogenetic trees, please visit our Phylogenetic Biology pages.
close boxIntroduction
Myctophids, or lanternfishes, are among the most abundant group of mesopelagic fishes in the World's oceans. They range from the Arctic to the Antarctic and, as a result of their diurnal migrations, can be found from the surface waters down to depths exceeding 2000 m. Although predominantly fishes of open oceanic waters, some species occur over continental slopes and even over continental and island shelves where they form components of coral reef communities. The family contains some 230-250 species arrayed within 30-35 genera (Paxton, 1972, 1979).
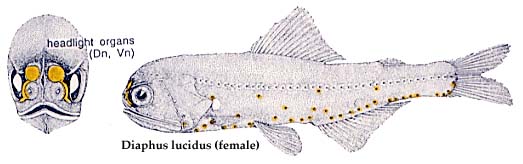
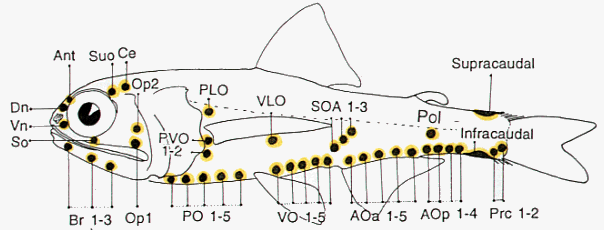
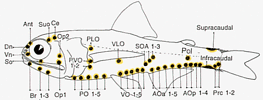
A noteworthy characteristic of the Myctophidae is the presence of non-bacterial luminescent organs (forming the lanterns of these so-called lanternfishes). Most prominent and widespread of the myctophid light organs are photophores. And with the exception of a single species (Taaningichthys paurolychnus, figured below) round, or keyhole shaped, photophores are found underlying modified scales on the head and ventral body surface of all myctophids.

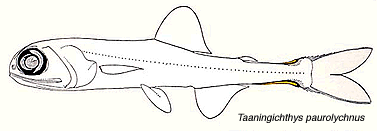
Taaningichthys paurolychnus is the only myctophid species lacking photophores. Interestingly all other members of the genus have these organs. Illustration courtesy of the Sears foundation for Marine Research, Yale University.
Most of these photophores are arrayed in highly characteristic and ordered patterns.

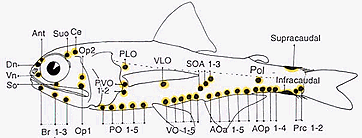
Schematic representation of myctophid luminous organs. The presence and placement of the photophore organs are important taxonomic characters at the generic and species levels. Drawing by M.L.J. Stiassny.
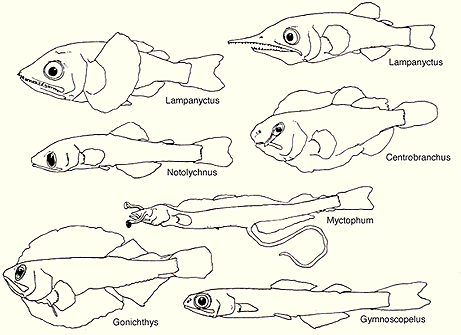
Variation in these basic patterns have long served as a basis for species diagnoses as well as in the development of classificatory schemes. Noteworthy in this regard, is the degree of sexual dimorphism in the arrangement, size and complexity of the accessory luminous organs of some myctophid taxa. For example, in Lobianchia the male bears a well-developed supracaudal luminous organ while the female lacks the supracaudal organ but bears a small infracaudal luminous organ.


Many myctophid genera exhibit marked sexual dimorphism of accessory luminous organs. Illustrations courtesy of Sears Foundation for Marine Research, Yale University.
Similarly, within the genus Diaphus there is often marked sexual dimorphism in the degree of development of their luminous "headlight" organs. These organs are usually larger and better defined in males than in females.
Marshall (1979) recalls an interesting observation made by William Beebe who found that an aquarium specimen ofMyctophum affine "replied" to the repeated illumination of his luminous watch dial with strong bursts of light from its caudal luminous organ. In contrast to the highly phasic output of the accessory luminous organs, body photophores emit a relatively steady glow of blue, green or yellow light of low intensity (Hulley, 1996). Excellent reviews of the comparative morphology, histology, operation, and possible functions of myctophid photogenic tissues are given by Edwards and Herring (1977) and Nafpaktitis et al. (1977).
The great majority (but by no means all) myctophid species undergo extensive vertical (diurnal) migrations and while average peak abundance during the day ranges between 300-1200 m, at night peaks are more usually between 10-100 m (at or around the surface mixing zone). Interestingly, while most species migrate some hundreds of meters into the trophically rich surface waters, not all individuals within a given population will do so every night. It seems that migratory disposition may depend on a complex of factors such as recency of last feeding, general condition, and reproductive state. Most myctophids are microcarnivores feeding on copepods, euphausiids, ostracods and amphipods and much of their trophic anatomy reflects the essentially planktivorous niche they occupy. Myctophids, in turn, are preyed upon by a variety of fishes and other marine vertebrates, most notably cetaceans.
Threats to myctophid populations
Worldwide, myctophids make-up at least 50% of all fish larvae taken in open-water plankton tows (Moser and Ahlstrom, 1974), and as adults they comprise some 65% of all mesopelagic fishes.

The diversity of myctophid larvae is noteworthy and larval characteristics have proven to be valuable markers for phylogenetic analysis (after Moser et al., 1984).
The economic potential of these fishes has been reviewed by GjØsaeter and Kawaguchi (1980), and they estimate global myctophid biomass to be around 600 million tonnes (a one hour trawl off the coast of Argentina is recorded as having yielded some 30 tonnes of a single myctophid species!). While few myctophids are currently considered palatable, with the worldwide decline in traditional marine fisheries, the family is of obvious potential for increased exploitation. In this regard a cautionary note seems in order - with such a large biomass undergoing regular vertical migration these animals play a pivotal role in oceanic energy dynamics, the overfishing of such a resource could have truly dire consequences.
Myctophid monophyly
While the intrarelationships of the various myctophid lineages are still a matter of debate (see below) there can be little doubt of the monophyly of the family. This fact was emphasized by Stiassny (1996) who found the family to be "bristling with synapomorphies" in just about every anatomical system that she investigated.
A selection of myctophid synapomorphies discussed by Stiassny (1996) includes, among other things, the presence of a medial row of toothplates arrayed along the length of the first branchial arch, maxillary control mediated via an A1-beta component of the adductor mandibulae muscle, a bony connection between the descending process of the third hypobranchial and the urohyal, and the reduction or loss of the dorsal hypohyal elements of the hyoid arch.
Phylogenetic Relationships
The most recent comprehensive analysis of myctophid intrarelationships is provided by Paxton et al. (1984). Their study is an abbreviated synthesis and reanalysis of a series of data collated from previous papers (Paxton, 1972; Moser and Ahlstrom, 1970, 1972, 1974; Moser et al., 1984). The Paxton et al. database contains 25 osteological characters, 17 larval characters, and 17 photophore characters assembled and coded in a matrix. However, their published tree was not derived from this matrix but instead was based on a "knowledge of the family" rather than a parsimony analysis of character distributions. Shortcomings in the Paxton et al. study were discussed by Stiassny (1996) who reanalyzed their original matrix as well as added additional morphological characters to produce a matrix-derived consensus tree. Stiassny's (1996) final tree recognizes the traditional two myctophid subfamilies as monophyletic; the Myctophinae (Metelectrona, Hygophum, Myctophum, (Benthosoma, Diogenichthys ), (Electrona (Kreftichthys, Protomyctophum )), (Symbolophorus (Loweina, Tarltonbeania ) (Gonichthys, Centrobranchus )), and the Lampanyctinae (Notolychnus (Bolinichthys, Triphoturus, Stenobrachius, Parvilux, Lampanyctus, (Lampadena, Taaningichthys ), (Ceratoscopelus, Lepidophanes ), (Lobianchia, Diaphus, Idiolychnus ), (Lampanyctodes, Gymnoscopelus, Hintonia, (Lamphichthys (Scopelopsis, Notoscopelus ))). Stiassny (1996) stressed that her analysis was extremely preliminary, and that considerably more work is necessary before the precise intrarelationships of the Myctophidae can be considered to be satisfactorily resolved. However, based on data in hand perhaps the finding of most interest is the resolution of the enigmatic, diminutive Notolychnus as the basal lampanyctine lineage.

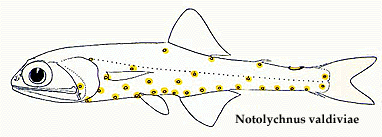
The phylogenetic placement of the monotypic genus Notolychnus has proven problematical but most recent workers concur that this taxon is the sister to the remaining lampanyctine radiation.
This result is in accord with Paxton's (1972) original (albeit highly trepidatious) placement of the taxon. Further resolution of myctophid intrarelationships remains an outstanding problem in teleostean systematics.
References
Edwards, A.S. and Herring, P.J. (1977). Observations on the comparative morphology and operation of the photogenic tissues of myctophid fishes. Mar. Biol. 41: 59-70.
Hulley, P.A. (1996). Laternfishes.:127-128. In. Encyclopedia of fishes. Paxton, J.R. and Eschmeyer, W.N. (eds). Academic Press.
GjØsaeter, J. and Kawaguchi, K. (1980). A review of the World's resources of mesopelagic fish. FAO Fisheries Tech. Pap. 193, 151 p.
Marshall, N.B. (1954). Aspects of deepsea biology. Hutchinson, London, 380 p.
Moser, H.G. and Ahlstrom, E.H. (1970). Development of laternfishes (family Myctophidae) in the California Current. Part. 1. Species with narrow-eyed larvae. Bull. Los Angeles Cty. Mus. Nat. Hist. 7: 1-145.
Moser, H.G. and Ahlstrom, E.H. (1972). Development of the laternfish, Scopelopsis multipunctatus Braier, 1906, with a discussion of its phylogenetic position in the family Myctophidae and its role in a proposed mechanism for the evolution of photophore patterns in lanternfishes. Fish. Bull. 70: 541-564.
Moser, H.G. and Ahlstrom, E.H. (1974). Role of larval stages in systematic investigations of marine teleosts: The Myctophidae, a case study. Fish. Bull. 72(2): 391-413.
Moser, H.G., Ahlstrom, E.H., and Paxton, J.R. (1984). Myctophidae: Development. In "Ontogeny and Systematics of Fishes" (H.G. Moser, W.J. Richards, D.M. Cohen, M.P. Fahay, A.W. Kendall, Jr., and S.L. Richardson, eds), Spec. Publ. No. 1: 218-239. American Society of Ichthyologists and Herpetologists, Lawrence, KS.
Nafpaktitis, B.G., Backus, R.H., Craddock, J.E., Haedrick, R.L., Robinson, B.H., and Karnella, C. (1977). Family Myctophidae. In "Fishes of the Western North Atlantic". Sears Found. Mar. Res., Mem. No. 1, Part 7: 13-265. Yale University, New Haven, CT.
Paxton, J.R. (1972). Osteology and relationships of the lanternfishes (Family Myctophidae). Bull. Nat. Hist. Mus. L.A. Cty. 13: 1-81.
Paxton, J.R. (1979). Nominal genera and species of lanternfishes (Family Myctophidae). Contrib. Sci., Nat. Mist. Mus. L.A. Cty. 322: 1-28.
Paxton, J.R., Ahlstrom, E.H., and Moser, H.G. (1984). Myctophidae: Relationships. In "Ontogeny and Systematics of Fishes" (H.G. Moser, W.J. Richards, D.M. Cohen, M.P. Fahay, A.W. Kendall, Jr., and S.L. Richardson, eds), Spec. Publ. No. 1: 239-244. American Society of Ichthyologists and Herpetologists, Lawrence, KS.
Stiassny, M.L.J. (1996). Basal ctenosquamate relationships and the interrelationships of the myctophifomr (scopelomorph) fishes. In "Interrelationships of Fishes" (Stiassny, M.L.J., Parenti, L.R. and G.D. Johnson, eds). Academic Press, San Diego, CA.
About This Page
Melanie L.J. Stiassny

American Museum of Natural History, New York, NY, USA
Page copyright © 1997
All Rights Reserved.
Citing this page:
Stiassny, Melanie L.J. 1997. Myctophidae. Lanternfishes. Version 01 January 1997 (under construction). http://tolweb.org/Myctophidae/15174/1997.01.01 in The Tree of Life Web Project, http://tolweb.org/




 Go to quick links
Go to quick search
Go to navigation for this section of the ToL site
Go to detailed links for the ToL site
Go to quick links
Go to quick search
Go to navigation for this section of the ToL site
Go to detailed links for the ToL site